Complete definition of how mutations affect antibodies used to prevent RSV
Jesse Bloom
Fred Hutch Cancer Center / HHMI

Study led by Cassie Simonich
Pediatrics Medical Fellow
Seattle Childrens / Fred Hutch
These slides: https://slides.com/jbloom/rsv-antibodies

Respiratory syncytial virus (RSV) is leading cause of infant hospitalization in USA
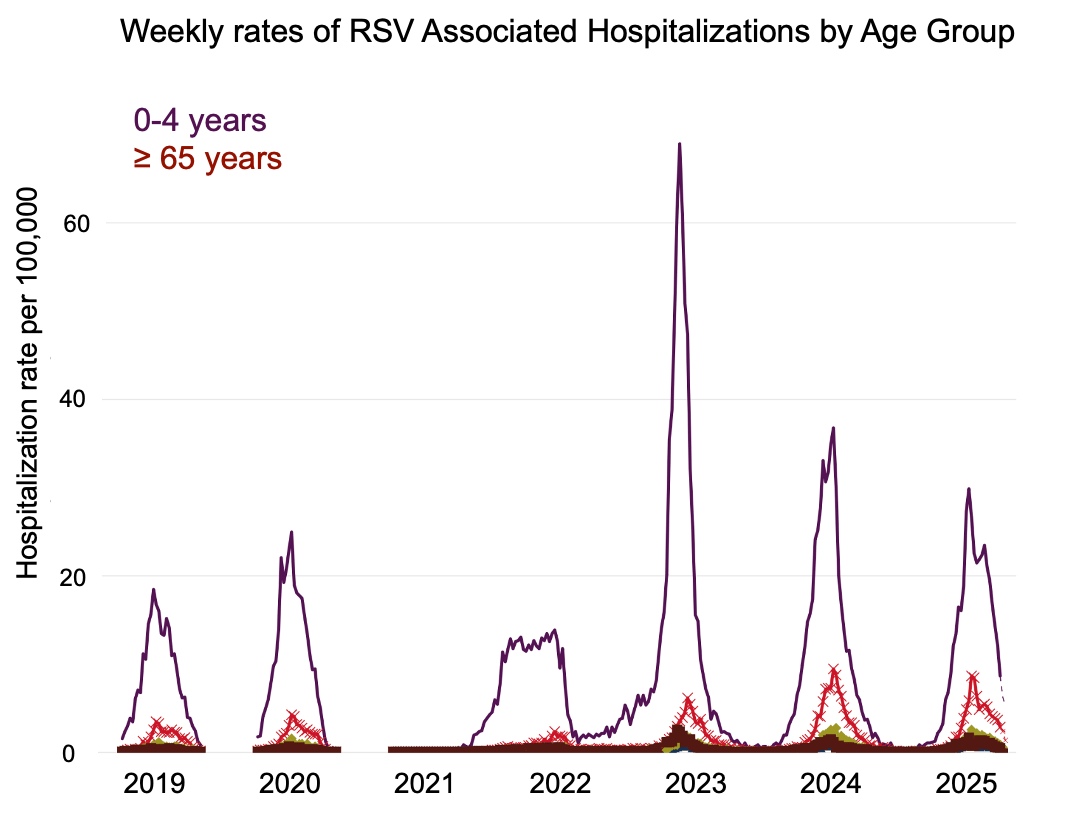
Plot from https://www.cdc.gov/rsv/php/surveillance/rsv-net.html; see also Suh et al (2022)
Especially in infants, RSV can cause airway inflammation and difficulty breathing
In developed world, infants hospitalized with RSV receive supportive care (eg, oxygen, ventilation, fluids) and usually recover (~0.1% in-hospital case-fatality rate)
In developing world where supportive care not available, RSV is a leading cause of infant mortality (~100,000 infant deaths per year)
Two approaches can help protect infants from RSV
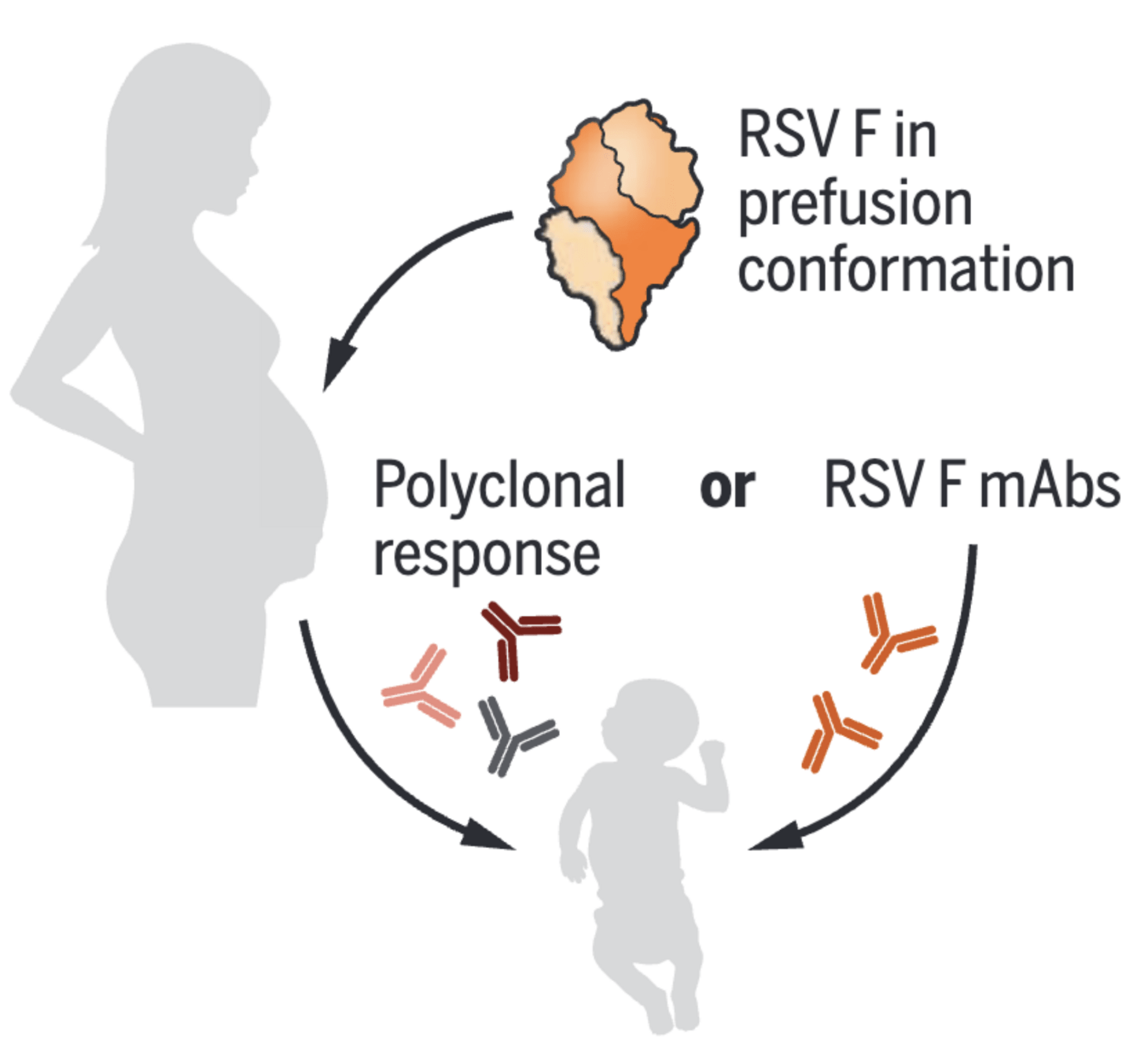
Direct administration of monoclonal antibody targeting RSV F protein
Maternal vaccination with prefusion stabilized RSV F protein

Direct administration of monoclonal antibody targeting RSV F protein
Maternal vaccination with prefusion stabilized RSV F protein
This talk focuses on anti-F monoclonal antibodies
RSV F fuses viral and cell membranes
Antibodies can bind to F and neutralize viral infection of cells
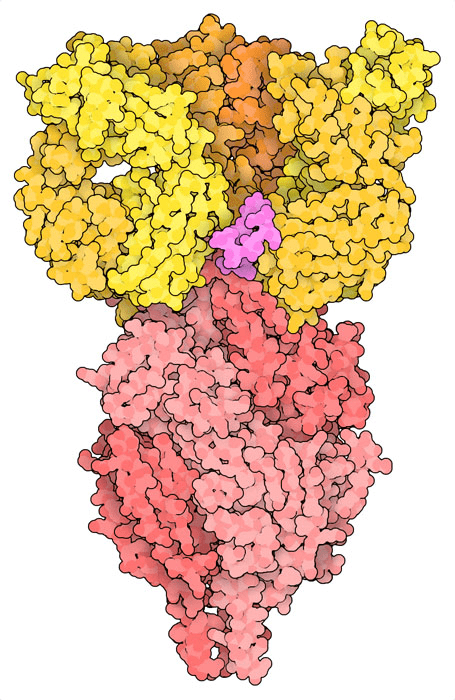
the antibody nirsevimab bound to prefusion F at epitope that includes glycan
Antibodies can bind to F and neutralize viral infection of cells

the antibody nirsevimab bound to prefusion F at epitope that includes glycan

Economic challenges of monoclonal antibodies against respiratory viruses
Although population disease burden is substantial, annual risk to any individual is low, and impossible to prospectively identify who will become severely ill at any given time.
Monoclonal antibodies are expensive to produce, and repeated dosing is required for sustained protection.
Viruses can evolve to become resistant.
Although population disease burden is substantial, annual risk to any individual is low, and impossible to prospectively identify who will become severely ill at any given time.
Monoclonal antibodies are expensive to produce, and repeated dosing is required for sustained protection.
Viruses can evolve to become resistant.
Why anti-RSV antibody prophylaxis for infants is economically feasible
Severe disease concentrated in an easily identifiable population (infants).
Although population disease burden is substantial, annual risk to any individual is low, and impossible to prospectively identify who will become severely ill at any given time.
Monoclonal antibodies are expensive to produce, and repeated dosing is required for sustained protection.
Viruses can evolve to become resistant.
Severe disease concentrated in an easily identifiable population (infants).
Infants require lower dose (they're smaller), most need protection only for first year, and progress has been made in engineering more potent and long-lived antibodies.
Why anti-RSV antibody prophylaxis for infants is economically feasible
Although population disease burden is substantial, annual risk to any individual is low, and impossible to prospectively identify who will become severely ill at any given time.
Monoclonal antibodies are expensive to produce, and repeated dosing is required for sustained protection.
Viruses can evolve to become resistant.
Severe disease concentrated in an easily identifiable population (infants).
Infants require lower dose (they're smaller), most need protection only for first year, and progress has been made in engineering more potent and long-lived antibodies.
This is a still a concern, and is topic of this talk.
Why anti-RSV antibody prophylaxis for infants is economically feasible
Brief history of anti-F antibodies for RSV prevention in infants

Palivizumab
Lower potency than subsequent antibodies.
First approved primarily for prophylaxis of high-risk infants (eg, born prematurely at <36 weeks), with dosing of 15 mg/kg each month for five months.
Due in part to cost, recommendation progressively narrowed: by 2014 only for infants born <29 weeks gestational age or <32 weeks with chronic lung disease
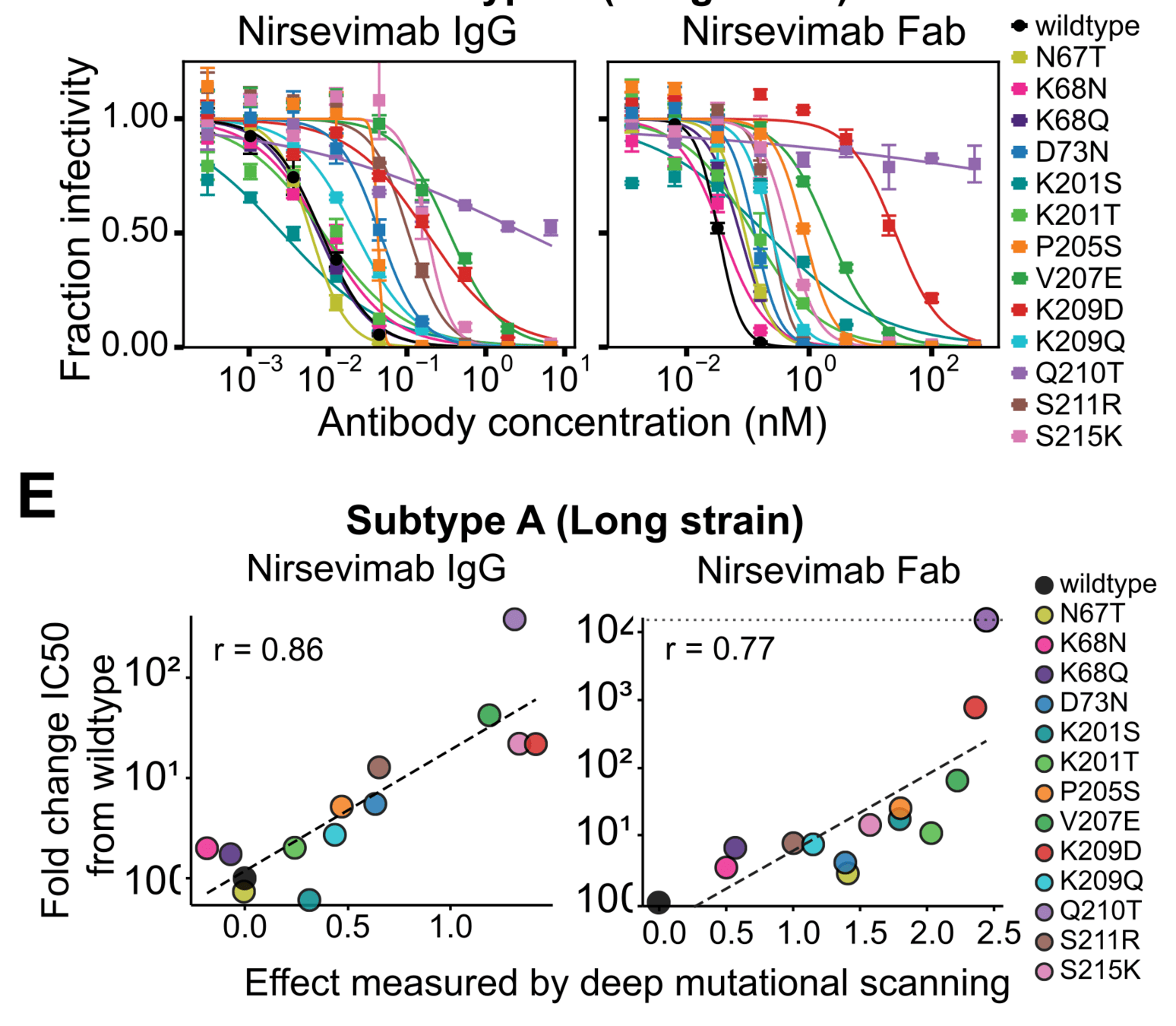
Neutralization curves from Simonich et al (2025)

Suptavumab
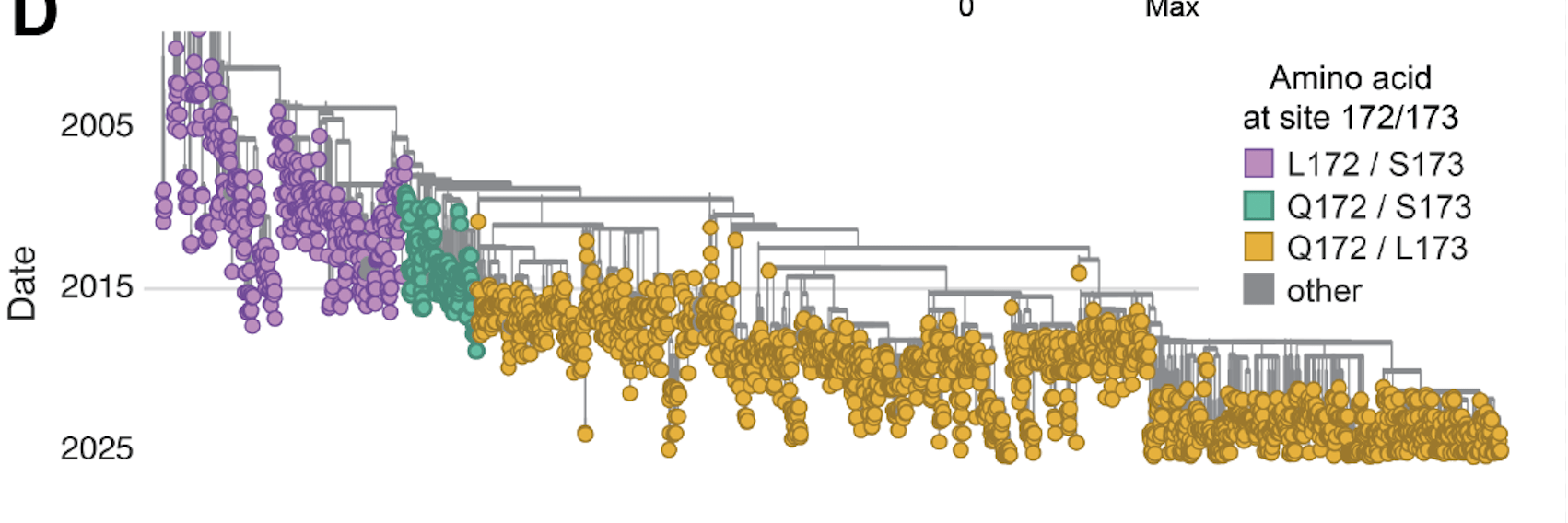
Developed by Regeneron: much more potent against some strains than palivizumab.
Failed Phase 3 clinical trial from 2015-2017 due to lack of efficacy against subtype B; coincided with evolution of new variants with mutations at F sites 172 and 173.
Neutralization curves from Simonich et al (2025)

Developed by AstraZeneca and Sanofi; much more potent than palivizumab and has extended half life.
Recommended in 2023 for all infants <8 months old entering their first RSV season. Dosing just one injection of 50 mg for infants <5 kg.
~80% effectiveness in preventing RSV hospitalization.
Nirsevimab
Neutralization curves from Simonich et al (2025)

Clesrovimab
Developed by Merck, and has high potency and extended half life similar to nirsevimab but targets different region of F.
Similar recommendations for use as nirsevimab, only recently approved (in 2025)
Neutralization curves from Simonich et al (2025)
Pseudoviruses to study how mutations to F affect antibody neutralization
We developed efficient pseudovirus system for measuring RSV neutralization
Transfection of RSV F (and G) along with plasmids expressing lentiviral proteins creates pseudotyped viral particles, which can only undergo a single round of cell entry and are not pathogens.
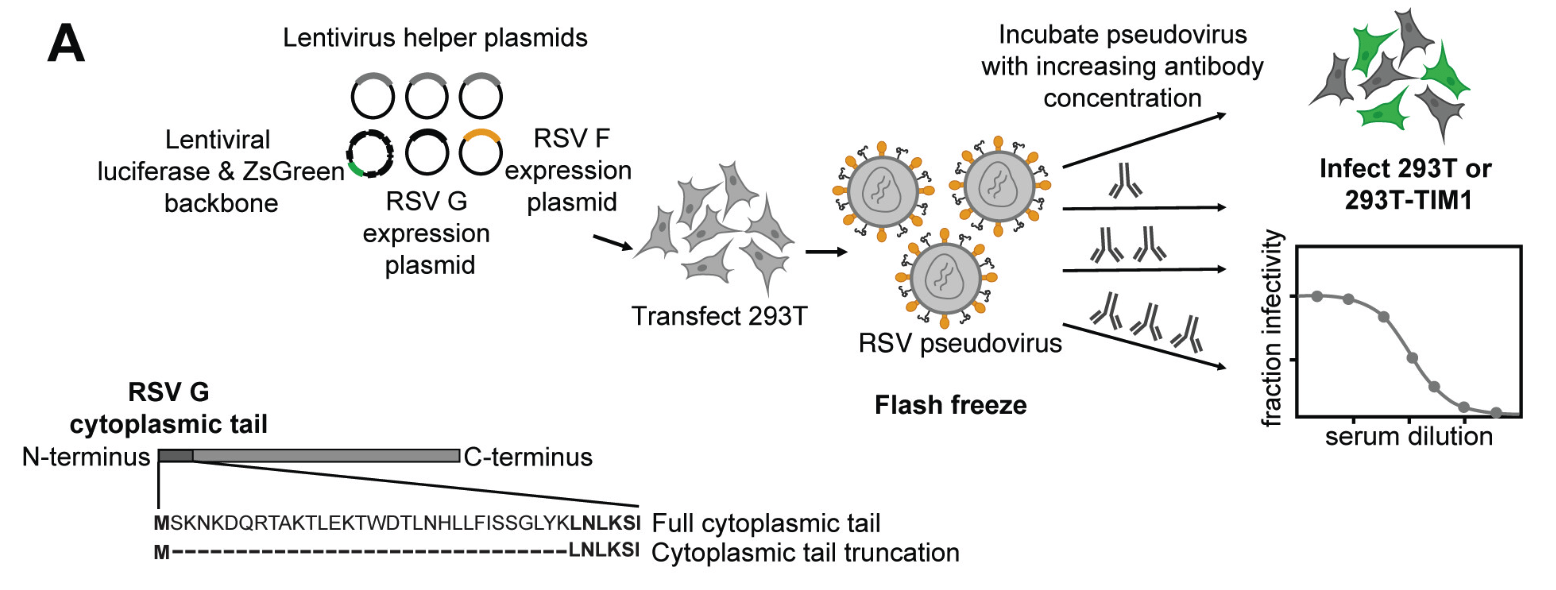
This system makes it possible to measure neutralization of any F mutant or variant

We can synthesize any variant of F and make pseudoviruses for neutralization assays, providing a safe way to study the effects of viral mutations on antibody neutralization.
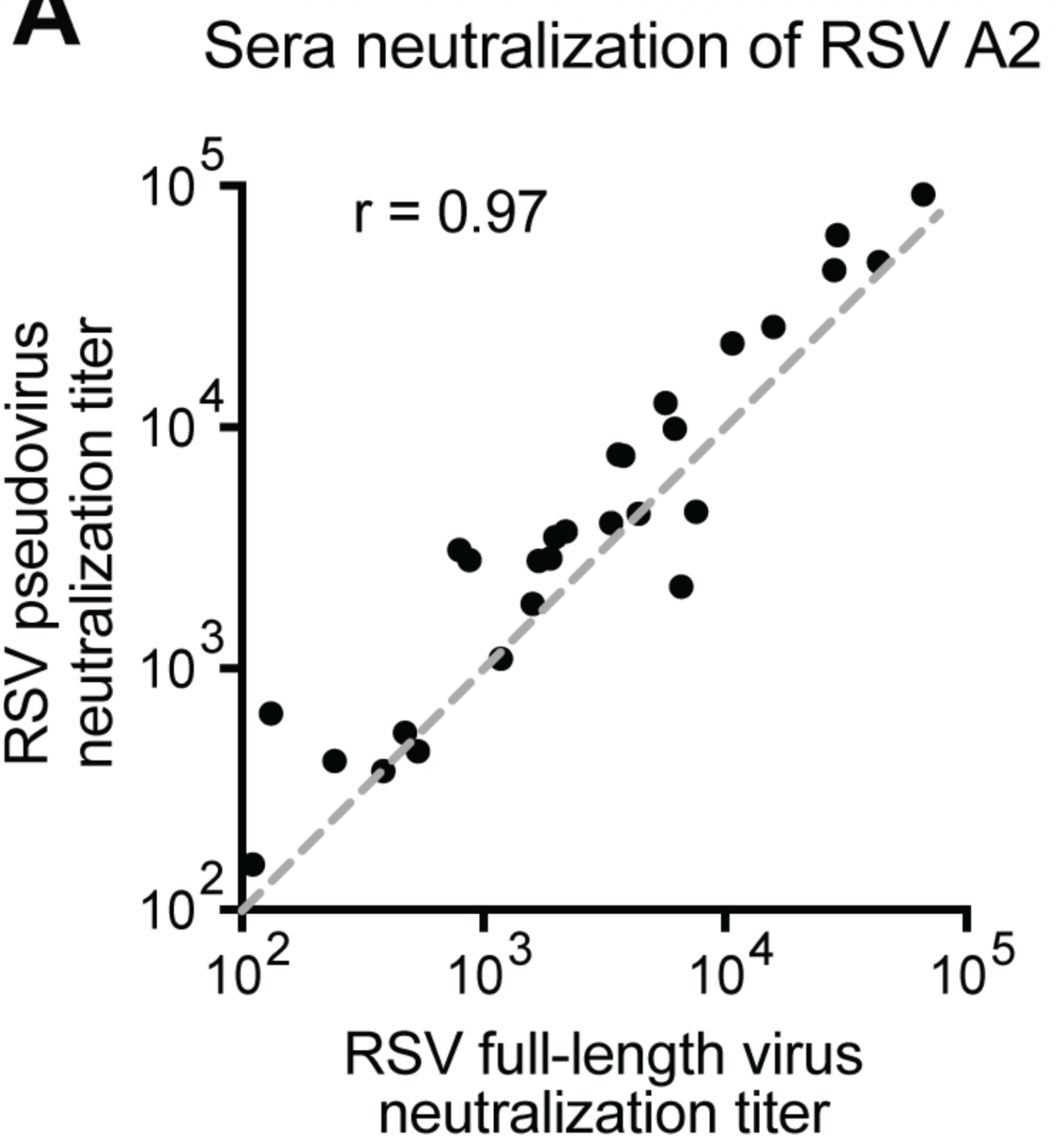
Neutralization measured by pseudovirus and authentic RSV is very similar
Titers of human sera against lab-adapted A2 strain of RSV measured by pseudovirus or live virus. For F from non-lab-adapted strains, pseudovirus approach much easier experimentally.
Impact of RSV evolution on polyclonal and monoclonal antibodies
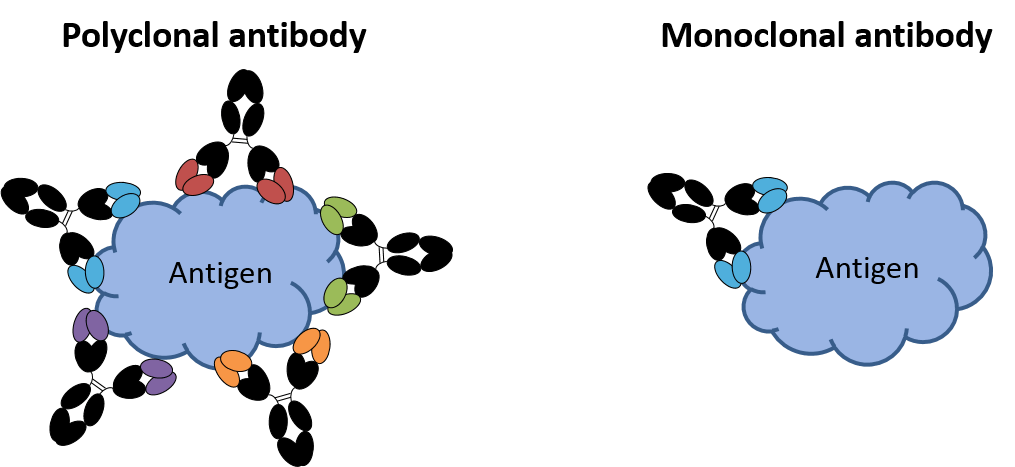
Infection induces polyclonal antibodies; current antibody prophylactics are monoclonal

Effect of RSV F evolution on human polyclonal antibodies
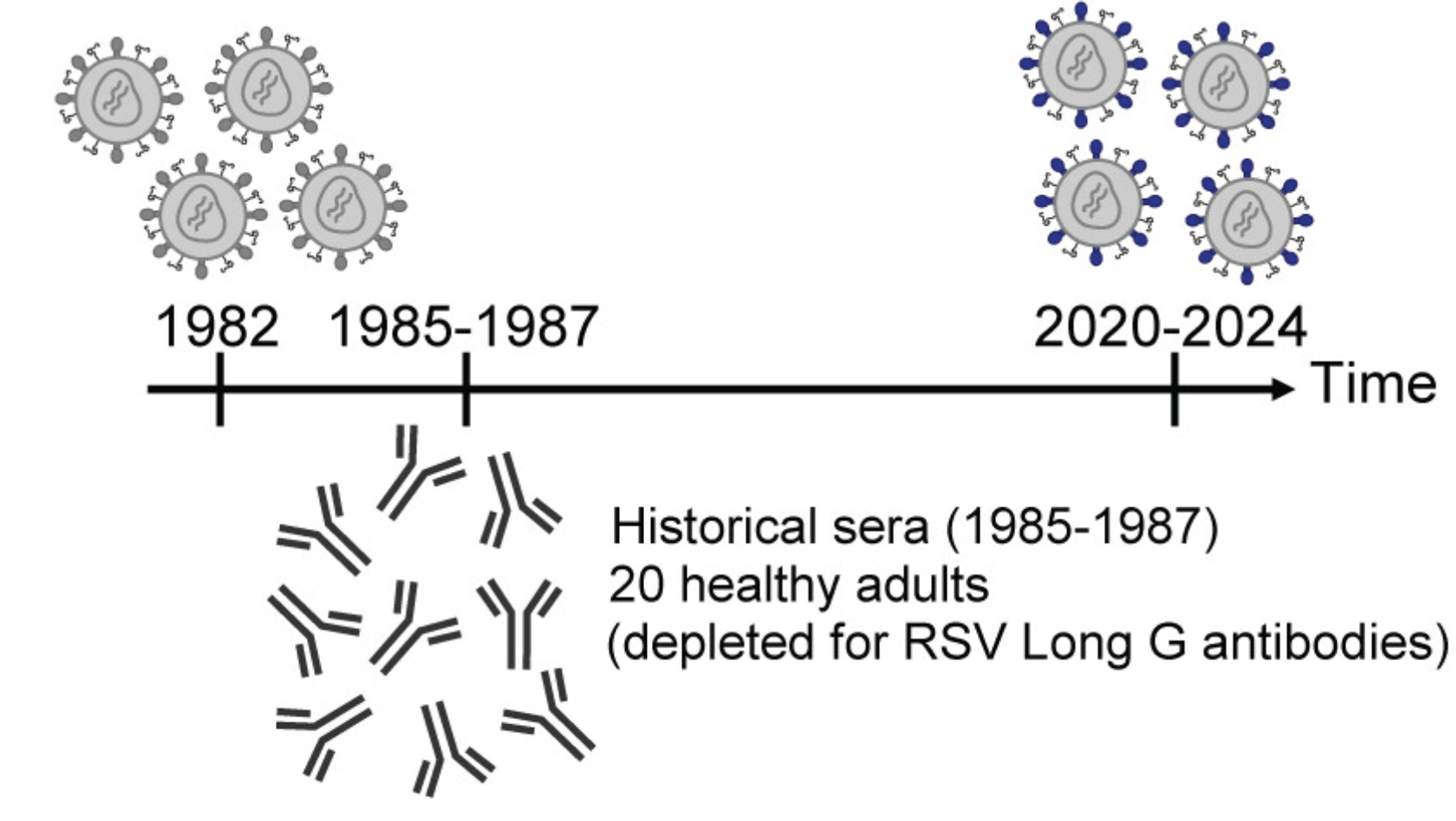
Measuring erosion of human polyclonal
antibody neutralization by viral evolution
Quantify ability of human sera collected in 1980s to neutralize older viruses that circulated in 1980s and recent/current viruses
If viral evolution erodes polyclonal antibody neutralization, titers lower to recent viruses


recent virus
serum neutralization titer
Hypothetical data
historical virus
Positive control: evolution of influenza hemagglutinin erodes polyclonal neutralization

1982 strain
2022 strain
serum neutralization titer
H3 influenza
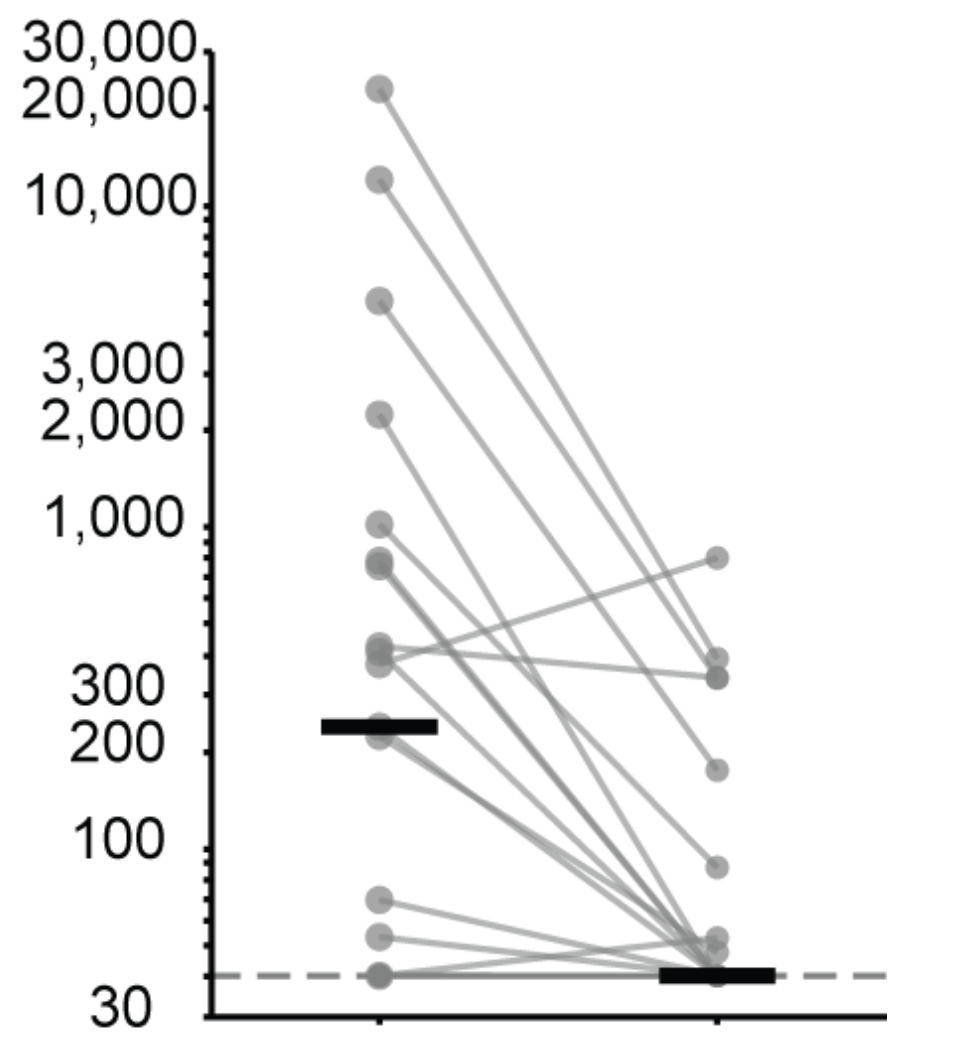
RSV F evolution does not strongly erode neutralization by human polyclonal antibodies

1982 strain
1992 strain
serum neutralization titer
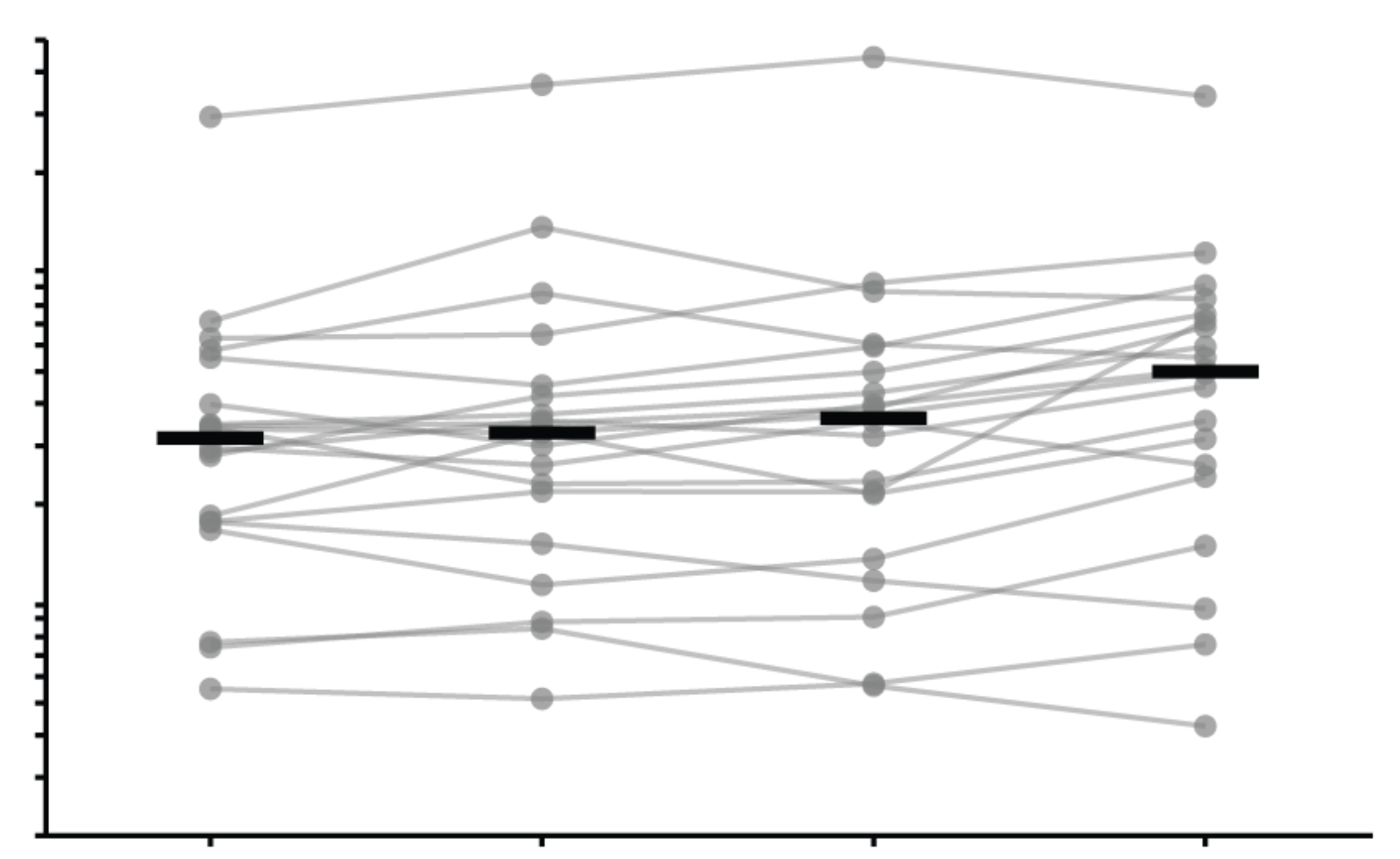
2019 strain
2024 strain
F from RSV subtype B

Effect of RSV F evolution on monoclonal antibodies

The clinical monoclonal antibody nirsevimab similarly neutralizes most RSV strains
plotted data from Simonich et al (2025)

Resistant strains have been identified in clinical and lab-passaging studies, plotted data from Simonich et al (2025)
But some (currently rare) natural RSV strains escape nirsevimab neutralization
Explaining the mysterious subtype-dependence of nirsevimab-escape mutations
Two subtypes of RSV: A & B. Their F proteins have ~90% sequence identity & similar structures
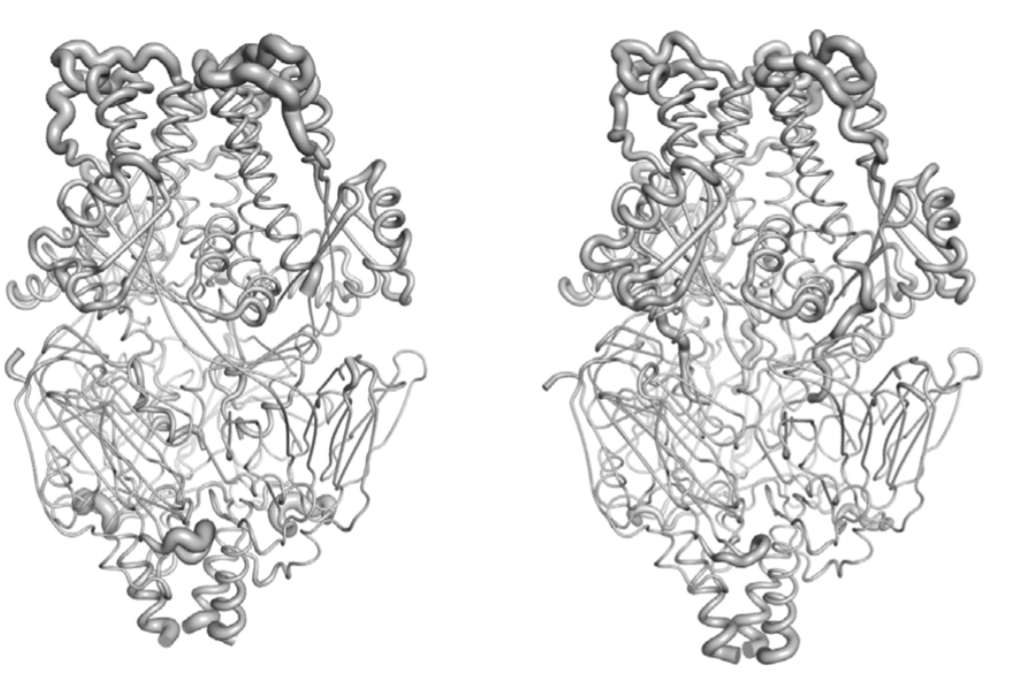
RSV B F structure
RSV A F structure
The structures have a RMSD deviation of only 1.8 angstroms.
Nirsevimab IgG has very similar neutralization activity against F from RSV A and RSV B
plotted data from Simonich et al (2025)

But resistance is more common for RSV B than A in infections of nirsevimab-dosed infants
| study | resistance in RSV-A | resistance in RSV-B |
|---|---|---|
| Fourati et al (2025a) | 2/195 = 1% | 23/184 = 13% |
| Fourati et al (2025b) | 0/236 = 0% | 2/24 = 8% |
| Ahani et al (2023) | 0/11 = 0% | 2/14 = 14% |
Rates of resistance to nirsevimab neutralization in RSV breakthrough infections of infants who received nirsevimab.
Experiments show some F mutations reduce nirsevimab neutralization of subtype B but not A

Why is RSV B more prone to nirsevimab resistance despite IgG neutralizing both subtypes similarly?
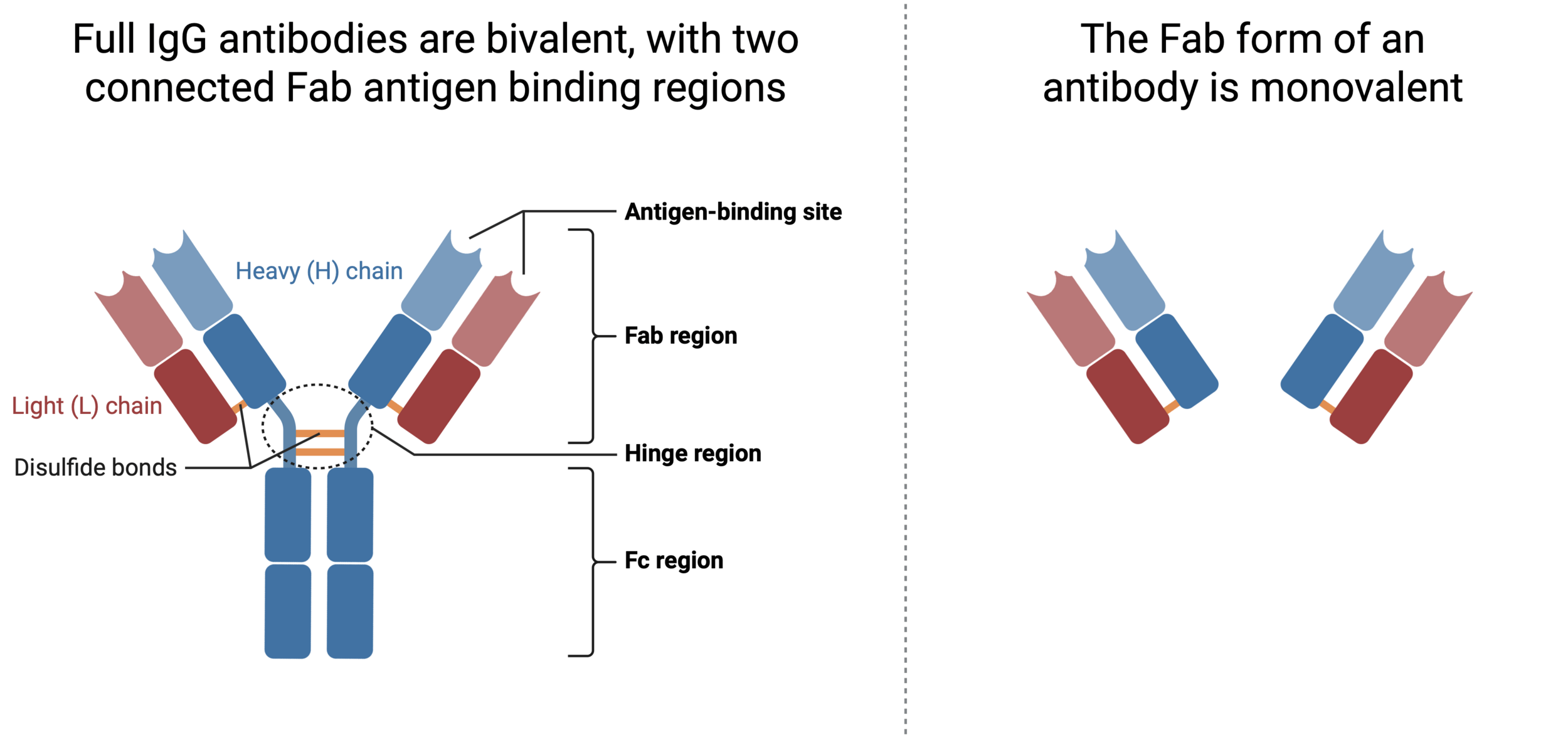
IgG antibodies (like nirsevimab) are bivalent, with two connected Fabs
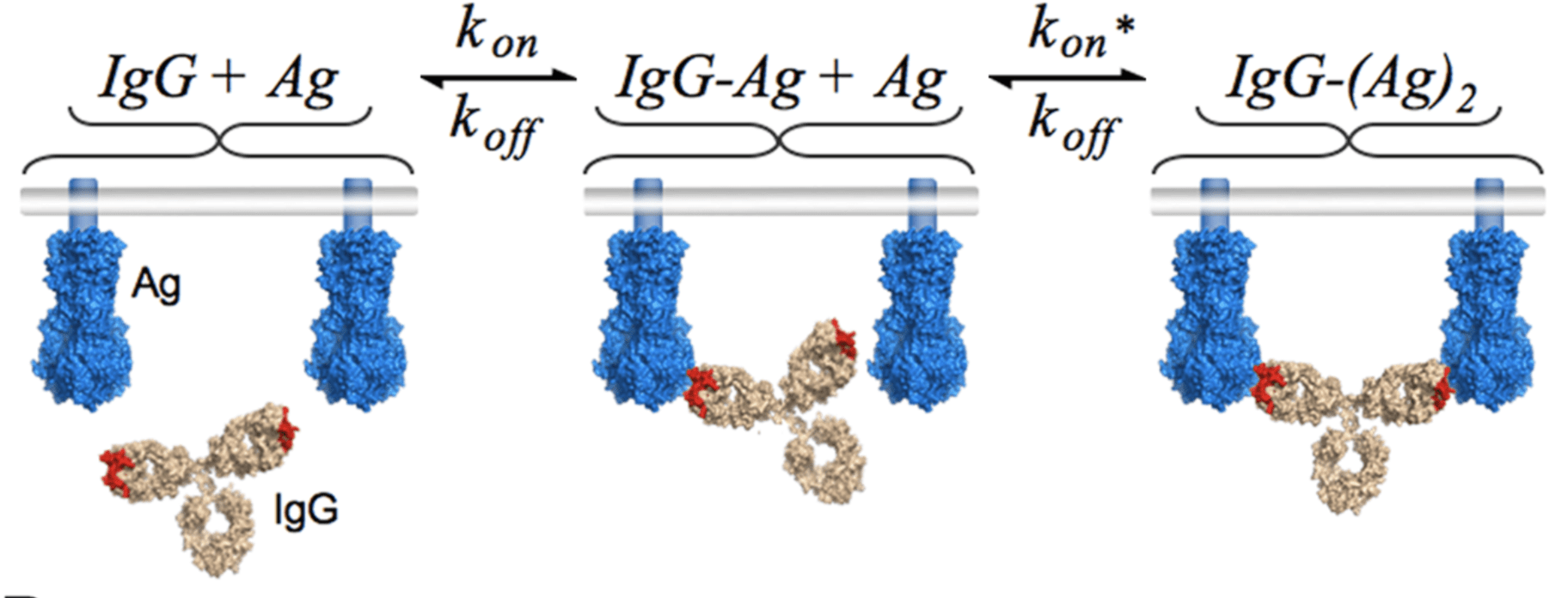
The apparent affinity (avidity) of bivalent IgG to antigen can be very high
In high avidity regime, viral mutations can reduce Fab but not IgG neutralization
See https://jbloomlab.github.io/IgG-vs-Fab-neutralization/notebook.html for full mathematical model
See https://jbloomlab.github.io/IgG-vs-Fab-neutralization/notebook.html for full mathematical model
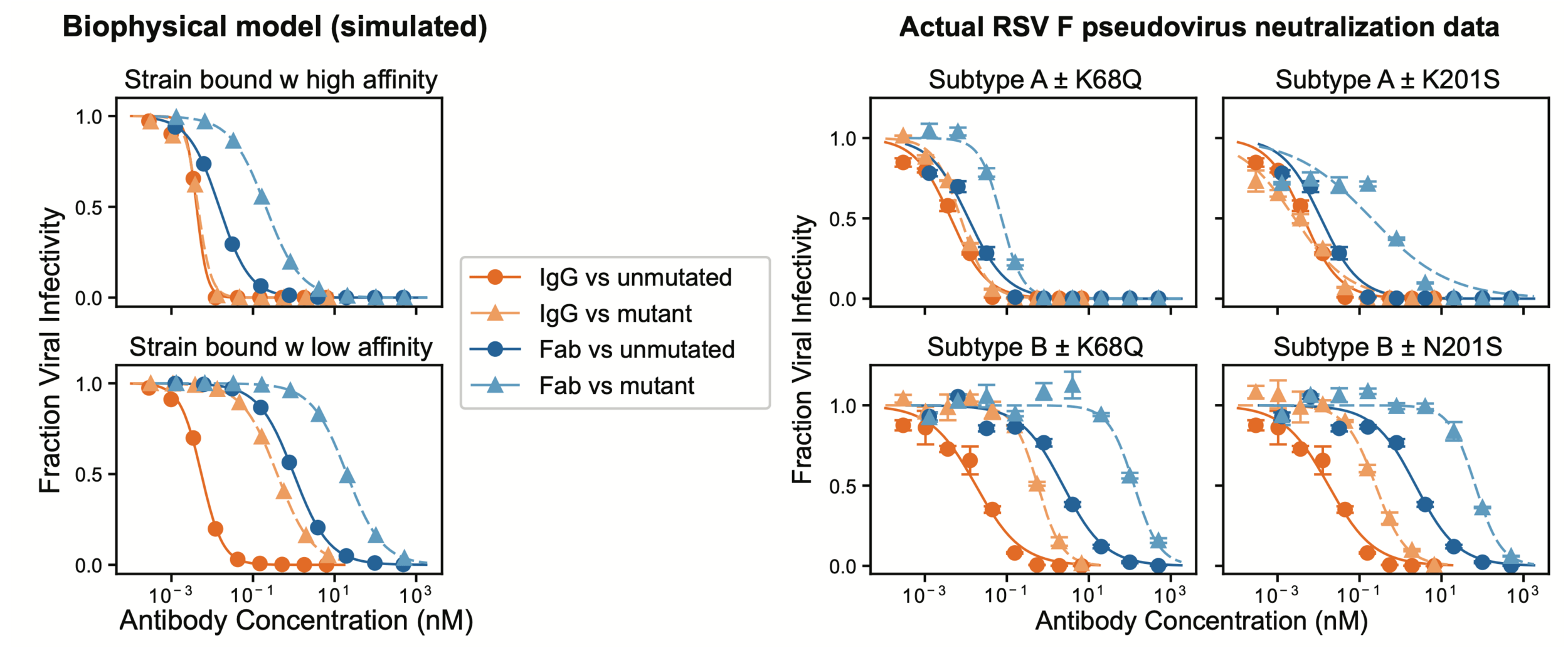
Model predicts viral mutations only reduce IgG neutralization of strain with lower Fab affinity

Some RSV F mutations reduce IgG neutralization of subtype B, but Fab neutralization of A & B
Reason some RSV F mutations escape nirsevimab only in subtype B is bivalent IgG buffering; mutations similarly affect Fab neutralization for both subtypes
Pseudovirus deep mutational scanning to enable informed surveillance for RSV antibody resistance
Previously, resistance mutations identified by viral passaging and characterizing clinical isolates
Example studies serial passaging RSV in presence of antibodies:
Example studies identifying resistance mutations in clinical infections:
These approaches identify just a fraction of the mutations that can actually affect antibody neutralization.
We used deep mutational scanning to quantify how all F mutations affect neutralization
Library of pseudoviruses expressing all single amino-acid mutants of RSV F.
Pseudoviruses can only undergo single round of cell entry, and so provide safe way to study effect of F mutations.
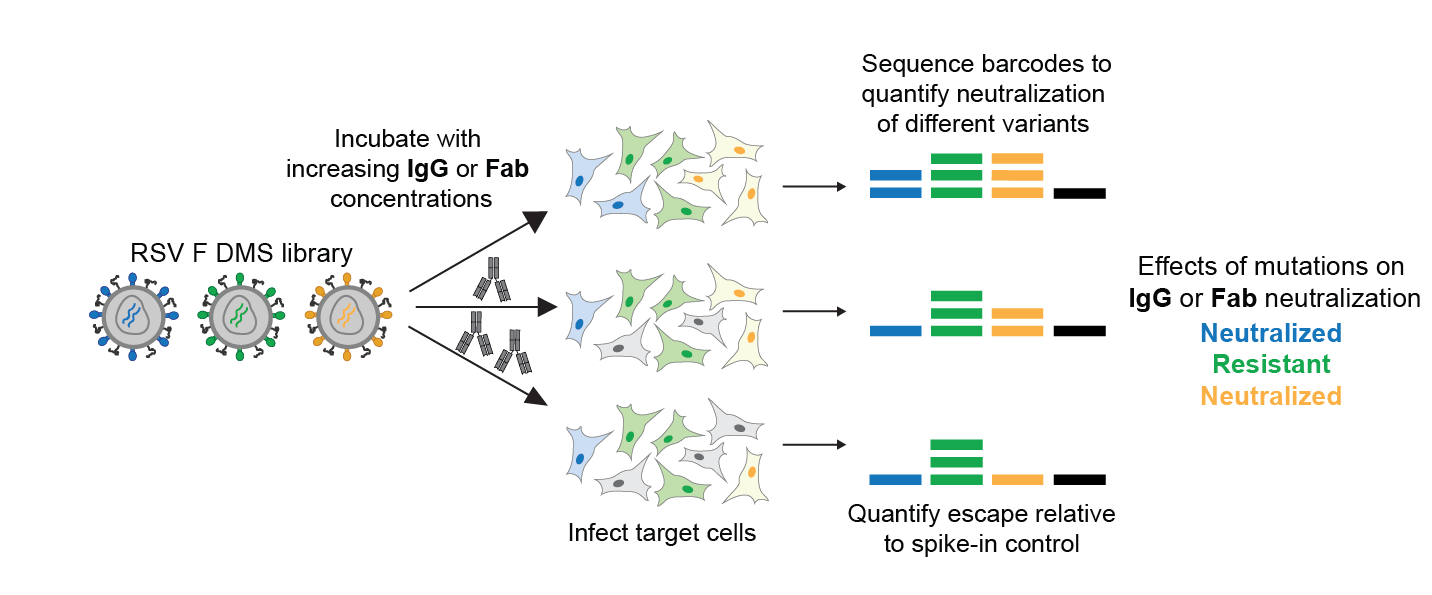
Workflow for measuring how all F mutations affect antibody neutralization
We can also measure how all mutations affect pseudovirus cell entry in absence of antibody, providing a measure of functional constraint.
How F mutations affect nirsevimab neutralization
Effects measured in deep mutational scanning correlate well with neutralization assays
How F mutations affect clesrovimab neutralization
Quantification of functional constraint on mutations that reduce nirsevimab and clesrovimab neutralization
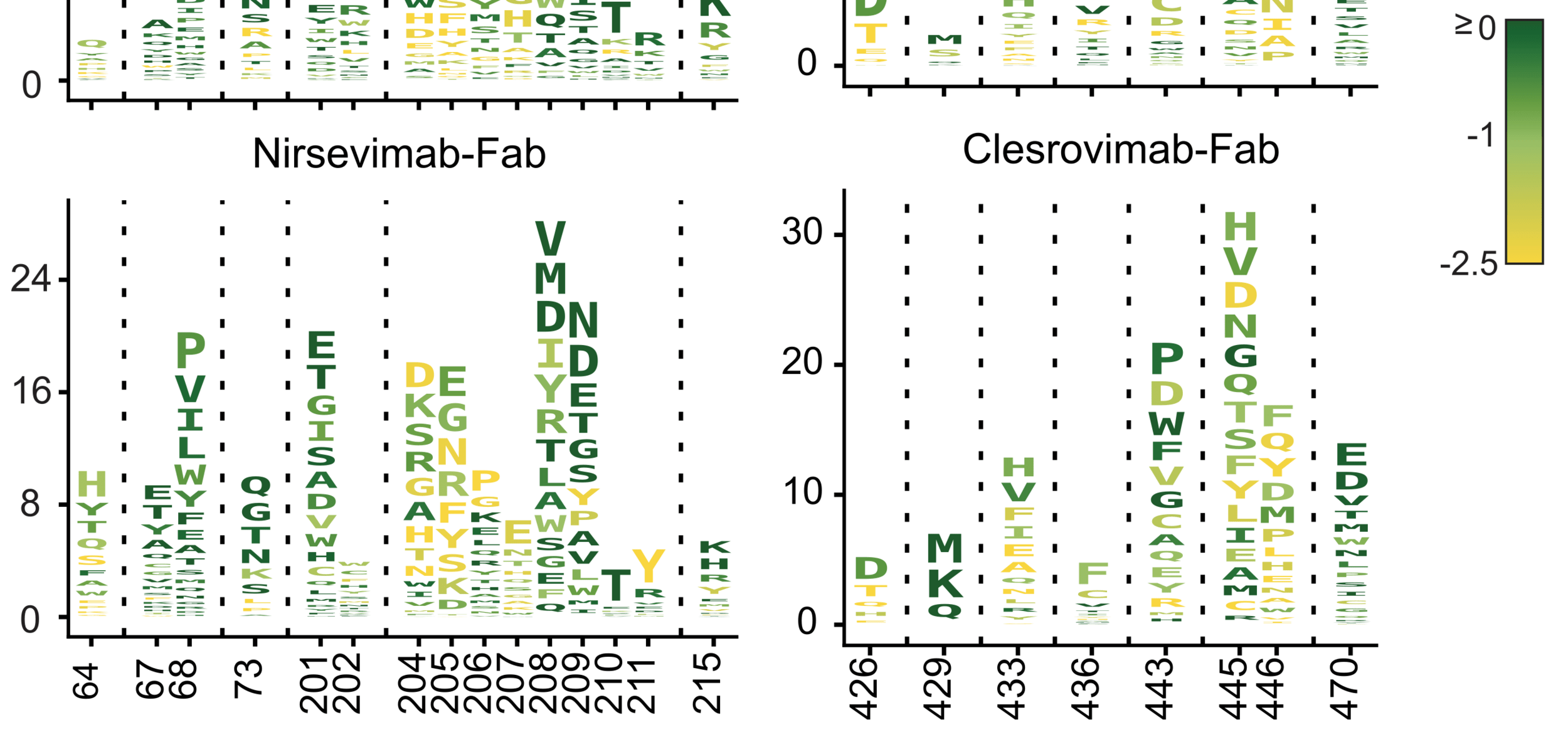

Letter heights indicate reduction in antibody neutralization, color indicates impact on F's cell entry function. These visualizations help quantify how constraint limits escape from different antibodies.
Nirsevimab
Clesrovimab
reduction in neutralization
site
cell entry function
We can use the deep mutational scanning data to score all natural F sequences for predicted neutralization
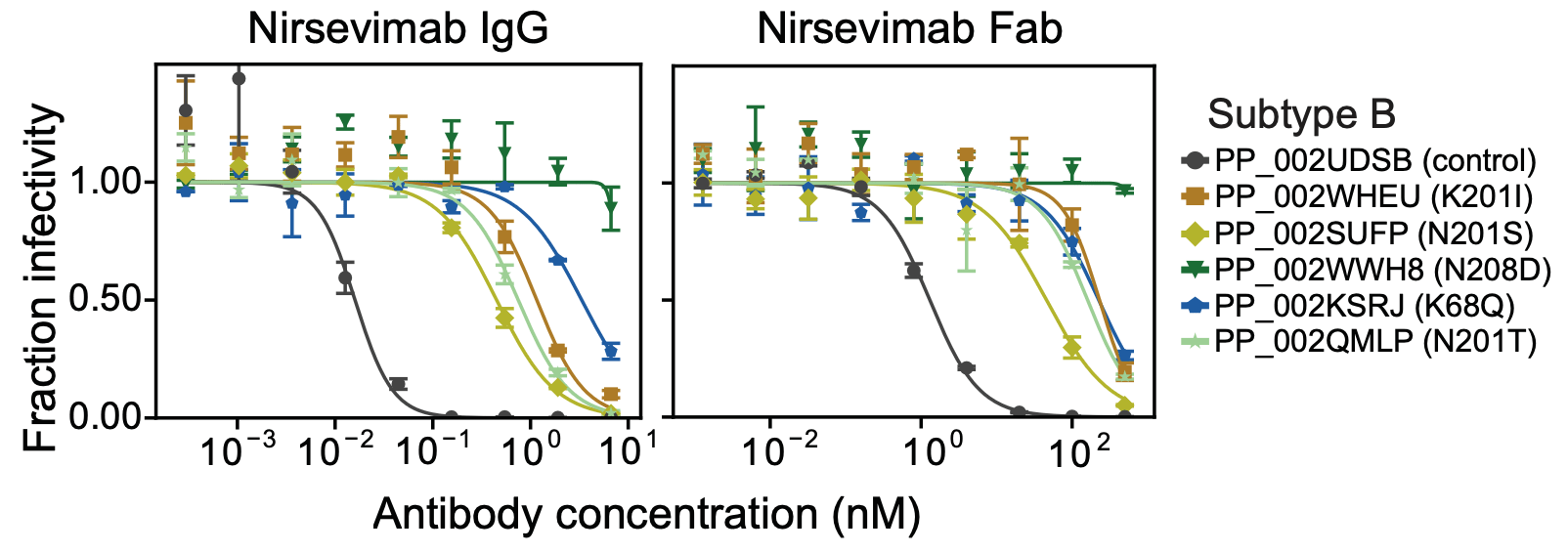
We can identify and validate natural strains with resistance

Our data therefore enable real-time surveillance for RSV antibody resistance
Conclusions
RSV antibodies have shown good effectiveness at preventing infant hospitalizations, but we need to be vigilant about potential resistance.
Difference in nirsevimab resistance between RSV subtypes A and B is due to bivalent IgG buffering of mutations that reduce Fab neutralization in subtype A but not B.
We have completely measured how RSV F mutations affect neutralization by antibodies in clinical use.
These data enable real-time surveillance for natural strains with resistance mutations.
All data are available for further analysis:
https://dms-vep.org/RSV_Long_F_DMS/
Thanks





Cassie Simonich
Pediatrics Medical Fellow
Seattle Childrens / Fred Hutch
Teagan McMahon
Research Technician
Fred Hutch

Lucas Kampman (Grad Student, Fred Hutch)
Helen Chu (University of Washington)
Richard Neher (University of Basel)
Alex Greninger (University of Washington)
These slides: https://slides.com/jbloom/rsv-antibodies